Abstract
-
Background
The current literature presents conflicting evidence regarding the effects of nonsteroidal anti-inflammatory drugs (NSAIDs) on fracture healing. This experimental study aimed to evaluate and compare the histological and immunohistochemical changes during femoral fracture healing in rats treated with a nonselective cyclooxygenase (COX) inhibitor (diclofenac sodium) and a selective COX-2 inhibitor (celecoxib).
-
Methods
Thirty-six male Wistar (standard outbred) albino rats weighing 200–400 g underwent standardized mid-diaphyseal femoral fracture surgery. The animals were randomized into three groups (n=12 per group): group 1 received diclofenac sodium, group 2 received celecoxib, and group 3 served as the control group and received 1 mL of distilled water orally once daily. Six rats from each group were euthanized at the end of the 2nd and 7th weeks after fracture for sample collection. Histological examination was complemented by immunohistochemical analysis, and the expression of type X collagen and vascular endothelial growth factor (VEGF) was assessed using the immunoreactive score (IRS) method.
-
Results
Healing scores were significantly higher in the control group at both time points (2nd week, P=0.01; 7th week, P=0.03). At the 2nd week, rats treated with diclofenac sodium demonstrated significantly greater fibrosis (P=0.01), and by the 7th week, they exhibited impaired bone formation (P=0.003) along with increased bone defects (P=0.01). IRS values for type X collagen and VEGF were significantly higher in the control group than in both treatment groups during the 2nd week (P=0.01 and P=0.005, respectively).
-
Conclusions
These findings suggest that, in this rat model, NSAIDs, particularly nonselective COX inhibitors, may disrupt the early phases of bone repair by affecting hypertrophic chondrocyte differentiation and reducing angiogenic activity. Although these results indicate a potential risk to optimal healing, they are preclinical observations, and their relevance to clinical fracture management should be interpreted with caution.
-
Level of evidence
V.
-
Keywords: Bone fractures, Non-steroidal anti-inflammatory drugs, Fracture healing, Cyclooxygenase inhibitors
Introduction
Background
Bone fractures represent one of the most prevalent public health challenges worldwide. In 2019 alone, approximately 178 million new fracture cases were reported globally, reflecting a 33.4% increase compared to previous years. Additionally, the total number of individuals living with fractures reached 455 million, marking a staggering 70.1% increase since 1990 [
1]. The region of Asia has a higher prevalence of individuals at high fracture risk with most cases being osteoporotic fractures in women [
2].
Fracture healing is a complex process that starts with the activation of immune and inflammatory responses. Macrophages, neutrophils, and platelets play a pivotal role in the initial phase of bone healing by releasing a wide array of pro-inflammatory cytokines and growth factors [
3]. These include tumor necrosis factor-alpha, platelet-derived growth factor (PDGF), bone morphogenetic proteins (BMPs), vascular endothelial growth factor (VEGF), transforming growth factor-beta, and various interleukins (IL-1, IL-6, IL-10, IL-11, IL-12, and IL-23), all of which are essential for initiating and regulating the healing cascade. These signaling molecules organize a cascade of cellular events that are essential for initiating and sustaining the healing process at the fracture site [
3-
5]. The two key factors on which successful fracture healing primarily depends are a favorable biological environment with sufficient vascularization and a mechanically stable environment [
6]. Previous studies have highlighted the central role of VEGF in fracture healing. VEGF promotes the development of a supportive vascular network that synergizes osteogenic factors such as BMP-2, and it drives the angiogenic responses necessary for cartilage resorption and subsequent bone formation. Enhanced vasculogenesis and angiogenesis increase the delivery of progenitor cells, nutrients, oxygen, and minerals essential for matrix deposition and mineralization. As repair progresses into later stages, chondrocytes within the cartilaginous callus cease proliferation, undergo hypertrophy, and begin producing type X collagen. These hypertrophic chondrocytes also express Osterix, a potent inducer of VEGF, which facilitates vascular invasion and the recruitment of chondroclasts into the hypertrophic cartilage [
7,
8]. However, several intrinsic and extrinsic factors can potentially disrupt or impair this healing process [
6]. One such factor influencing fracture healing is the use of nonsteroidal anti-inflammatory drugs (NSAIDs), which are frequently prescribed for their analgesic and anti-inflammatory properties [
9]. Despite their widespread use in clinical practice, the impact of NSAIDs on bone healing remains controversial. Numerous studies have investigated their effects, with findings ranging from delayed or impaired healing to no significant impact. The variability in results is often attributed to differences in the type of NSAID used, dosage, duration of administration, and the specific stage of healing during which the drug is administered. This ongoing debate underscores the need for cautious consideration when prescribing NSAIDs in the context of bone fractures [
10].
NSAIDs are often preferred over other analgesics in the management of fractures due to their dual action providing both effective pain relief and anti-inflammatory benefits [
11]. However, prolonged administration of NSAIDs has been associated with an increased risk of delayed union, malunion, and nonunion of fractures. In contrast, some studies have reported no significant impact on bone healing, leaving the role of NSAIDs in fracture repair a subject of ongoing debate [
9].
It is believed that due to the inhibition of cyclooxygenase (COX) enzymes by NSAIDs, the synthesis of prostaglandins is reduced which participate in different processes of healing and repair leading to repressive effect on fracture healing. Prostaglandins promote the process of angiogenesis and collagen synthesis thus enhancing the healing process [
10]. Based on the current literature regarding NSAID exposure and bone healing, there is a clear need for further research to better elucidate the mechanisms of action of different classes of NSAIDs and their specific effects on the various phases of fracture repair [
9].
In the current study we hypothesized that NSAID administration reduces collagen type X (COLX) and VEGF expression during fracture healing. This was investigated through detailed histological analysis and by determining the proportion and localization of COLX and VEGF in healing fractures.
Methods
Ethics statement
This study was conducted following approval from the Ethical Review Committee of the institute (approval No. UHS/Reg-21/ERC/510) and in accordance with institutional guidelines for medical and biomedical animal research.
Study design
Standard outbred Wistar albino rats were used as a fracture model in the current study as a sequence of biological events during fracture healing in these rats such as inflammation, callus formation, endochondral ossification, and remodeling closely parallel human fracture healing [
12].
Sample size was determined using the resource equation approach, which is appropriate for experimental animal studies where prior effect size estimates are limited. Based on this method, the minimum required sample size was calculated to be eight animals per group to achieve an adequate degree of freedom for error. To further increase statistical power and improve the detection of biologically meaningful differences, the sample size was increased to twelve animals per group. This calculation was based on the total number of animals per group and was not dependent on subdivision by time points; despite postoperative losses, the overall sample size per group remained above the calculated minimum requirement. Accordingly, a total of thirty-six healthy adult male Wistar albino rats (200–400 g) were randomly allocated to three groups using a lottery method.
To minimize potential confounders, the animals were kept separately in the institutional animal facility in small groups of 2–3 adult rats per cage under standardized conditions, including a well-ventilated environment maintained at a temperature of 23±2 °C, relative humidity of 55%±5%, and a 12-hour light/dark cycle. Cage location was not systematically varied; all animals were provided with a standard laboratory rodent diet and had free access to water. Surgical procedures, drug administration, and outcome assessments were conducted according to a standardized protocol and in the same order across groups where feasible. Any sick rats found during the research procedure were excluded from the study, in addition, animals who underwent any surgical complications or died both during and after surgery were also excluded from the study.
Surgical procedure and induction of fracture
Rats were given antibiotic enrofloxacin (5–10 mg/kg) prophylactically just before the surgery and anesthetized with mixture of Ketamine 80–100 mg/kg and Xylazine 5–10 mg/kg given intramuscularly [
13]. A vertical midline incision was made under aseptic and sterile conditions, beginning from the mid-thigh and extending over the knee joint, positioned medial to the patella and the extensor mechanism, on either the right or left limb as required. The quadriceps and extensor mechanism were displaced laterally. The distal end of the femur and the proximal end of the tibia were exposed after subluxation of the patella and extensor mechanism laterally. An entry hole in the medullary canal was created in the middle of the groove between the condyles of the femur [
13]. Intramedullary reaming was done with the passage of a guidewire. The mid-diaphyseal fracture of femur was induced by a surgical hammer and retrograde intramedullary nailing was done (
Supplementary material). The skin was closed in layers with the help of a Vicryl suture, postoperatively, the animals were monitored constantly until full recovery from anesthesia, followed by hourly observations for the first 8 hours postoperatively. Therapeutic dose of tramadol (10 mg/kg body weight) was administered intramuscularly once after surgery to all animals to keep them pain free [
14].
The animals were randomly divided into three groups using a lottery method, each comprising 12 rats. Group 1 received a nonselective NSAID (diclofenac sodium) administered once daily at a dosage of 5 mg/kg body weight/day [
15], while group 2 was treated with a selective COX-2 inhibitor (celecoxib) at a dosage of 4 mg/kg body weight/day [
16]. Both drugs were given orally and started from the first postoperative day. Group 3 served as the control group and was administered 1 mL of distilled water orally once daily (
Table 1).
Animals were monitored daily by trained personnel for behavioral signs of pain or distress, including reduced mobility, guarding behavior, altered grooming, and changes in food or water intake. A predefined rescue analgesia protocol and humane endpoints were established; however, no animal required additional analgesic intervention or early euthanasia during the study period.
Additional postoperative analgesics were not administered to the control group, as two of the experimental groups were already receiving NSAIDs. This approach was adopted to avoid potential confounding effects of additional pharmacological agents on bone healing outcomes and to minimize bias related to drug interactions.
Dissection and radiograph
Half of the animals from each group were euthanized and dissected at the end of the 2nd week (15th postoperative day). The other half of the animals were sacrificed at the end of 7th week (49th postoperative day). The dissected limbs of animals sacrificed at 7th week were radiographed laterally to evaluate the healing of bone.
Histological and immunohistochemical evaluation
The fractured femur was fixed in 10% formalin for 72 hours and then decalcified in 20% ethylenediaminetetraacetic acid solution (pH 8) for 4–5 days in a vacuum oven and paraffin blocks were made after tissue processing. Alcian blue hematoxylin/orange G (ABH/OG) staining was done on 5 µm thick sections to assess the healing bone based on modified Lane and Sandhu method [
17]. This method scores from 0–10 each for newly formed bone, newly formed cartilage, evidence of hypertrophic cartilage, newly formed fibrous tissue, and remnant defect size. Scoring increased with increased newly formed bone, cartilage and hypertrophied cartilage, while it decreased with an increase of fibrous tissue and bone defect between fractured ends.
ABH/OG-stained sections are used to appreciate architecture of the callus, as cartilage stains blue, immature bone gray, mature bone orange, erythrocytes and soft tissue appear pink to red and fibrous connective tissue violet.
Immunohistochemical analyses of COLX and VEGF were also done on 3 μm sections using polyclonal primary antibody (PA5-115039 and ABCLONAL A12303 respectively). The intensity and percentage expression were analyzed based on immunoreactive scoring system [
18]. In brief, immunoreactivity was evaluated using the immunoreactive score (IRS), calculated by multiplying the percentage of positive cells (0–4) by staining intensity (0–3). The resulting IRS values were categorized as: 0–1, negative; 2–3, mild; 4–8, moderate; and 9–12, strongly positive.
For histological and IRS scoring reliability analysis, the first two authors performed independent evaluations of radiographs and slides (for histology and immunohistochemistry). They also performed the same analysis twice on radiographs and slides by blinding the group allocation with one-month interval to assess intraobserver variation. Both inter and intraobserver agreement of each radiographic finding, histological manifestation and the total score were statistically analyzed using intraclass correlation coefficients (ICC), which demonstrated good to excellent agreement (ICC, 0.7–0.9).
Statistical analysis
Data were analyzed using IBM SPSS ver. 26.0 (IBM Corp.). Quantitative variables were summarized as median with interquartile range due to nonnormal data distribution. Normality was assessed using the Shapiro–Wilk test. Comparisons among the three groups for histological and immunohistochemical parameters were performed using the Kruskal-Wallis test, followed by Dunn’s post hoc test for pairwise comparisons where appropriate. Correlations between numerical variables were evaluated using Spearman’s rank correlation coefficient. Inter- and intraobserver reliability were assessed using intraclass correlation coefficients. A P-value ≤0.05 was considered statistically significant.
Results
General health of the animal during experimental period
Two animals (one from each diclofenac and control groups) died on the first postoperative day without any obvious reason from the sample cohort of 36 animals leaving a total of 11 animals in diclofenac and control group while 12 in celecoxib. During daily morning and afternoon inspection of animals to monitor health, movement, and food intake behavior, two animals of the same cage were found to have minor wounds at 14th day on their nonoperative leg, due to fighting and injuring each other, which was taken care of by antiseptic dressing. Throughout the remaining experimental period, these animals stayed healthy, no further sickness, disease, or change in behavior was noticed.
Radiographic findings
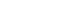
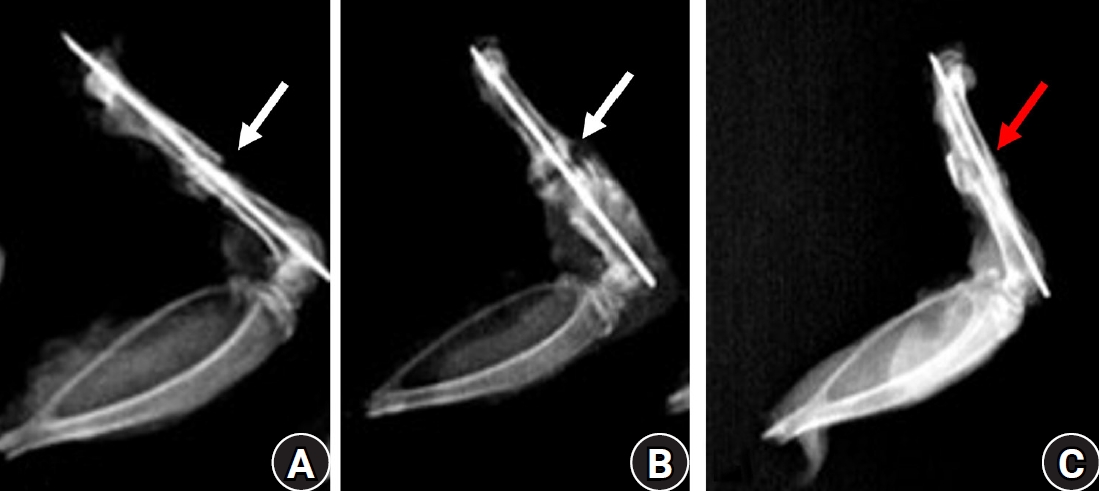
The radiographs of nearly 90% of the animals treated with diclofenac and 65% of those treated with celecoxib by the end of 7th week postoperative still showed the gap between the two edges of the healing bones and were marked as persistent radiolucent fracture lines by two observers. In contrast, all animals in the control group exhibited well-formed bony callus (
Fig. 1).
In ABH/OG-stained sections the cartilage, immature and mature bone and fibrous connective tissue were appreciated quite well (
Fig. 2). Lightly stained newly formed bone was seen surrounding the fractured ends. Bone defects appeared as tissue gap with cluster of newly formed chondrocytes present between and around fracture ends forming soft callus. Fibrosis was identified as groups of small fibroblasts present at the periphery of fractured ends and soft callus. Mature hypertrophied chondrocytes were observed as giant cells with large nucleus present on the junction of cartilage and newly formed bone tissue. The inter and intra observer ICC for all the histological parameters was excellent to fair.
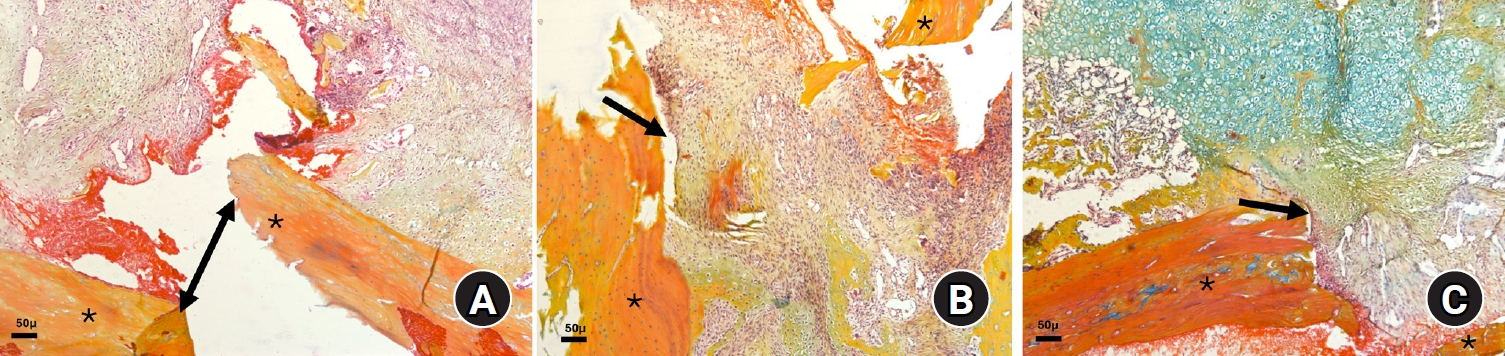
Among individual parameters, fibrosis scores were significantly lower in the NSAID-treated groups (P=0.03) indicating greater fibrous tissue at the fracture site. Pairwise comparison using Dunn’s post hoc test showed significant difference only between diclofenac group and controls (P=0.01). The overall healing scores were also found significantly lower in experimental animals (P=0.01) while pairwise comparison using Dunn’s post hoc test showed significant difference only between diclofenac group and controls (P=0.004) (
Table 2,
Fig. 3).
At the 7th week, newly formed bone was significantly greater in the control group compared with the experimental groups (P=0.01). Pairwise comparisons using Dunn’s post hoc test demonstrated a significant difference between the diclofenac and control groups (P=0.003). Bone defect scores were significantly lower in the diclofenac group (P=0.03) reflecting larger residual bone defects). Dunn’s post hoc analysis revealed a highly significant difference between the diclofenac group and controls (P=0.01), as well as borderline significance between diclofenac and celecoxib groups (P=0.05) (
Fig. 4). Total healing scores were significantly lower in experimental animals (P=0.03), with pairwise comparisons showing significant differences between the diclofenac group and controls (P<0.001) and between diclofenac and celecoxib groups (P=0.03) (
Table 2).
The immunohistochemical stained slides gave the variable expression of collagen X (COLX) and VEGF. The COLX staining was seen around the mature hypertrophied chondrocytes and near the newly formed bone, while VEGF expression was mostly near the newly formed bone, in extracellular matrix and around blood vessels.
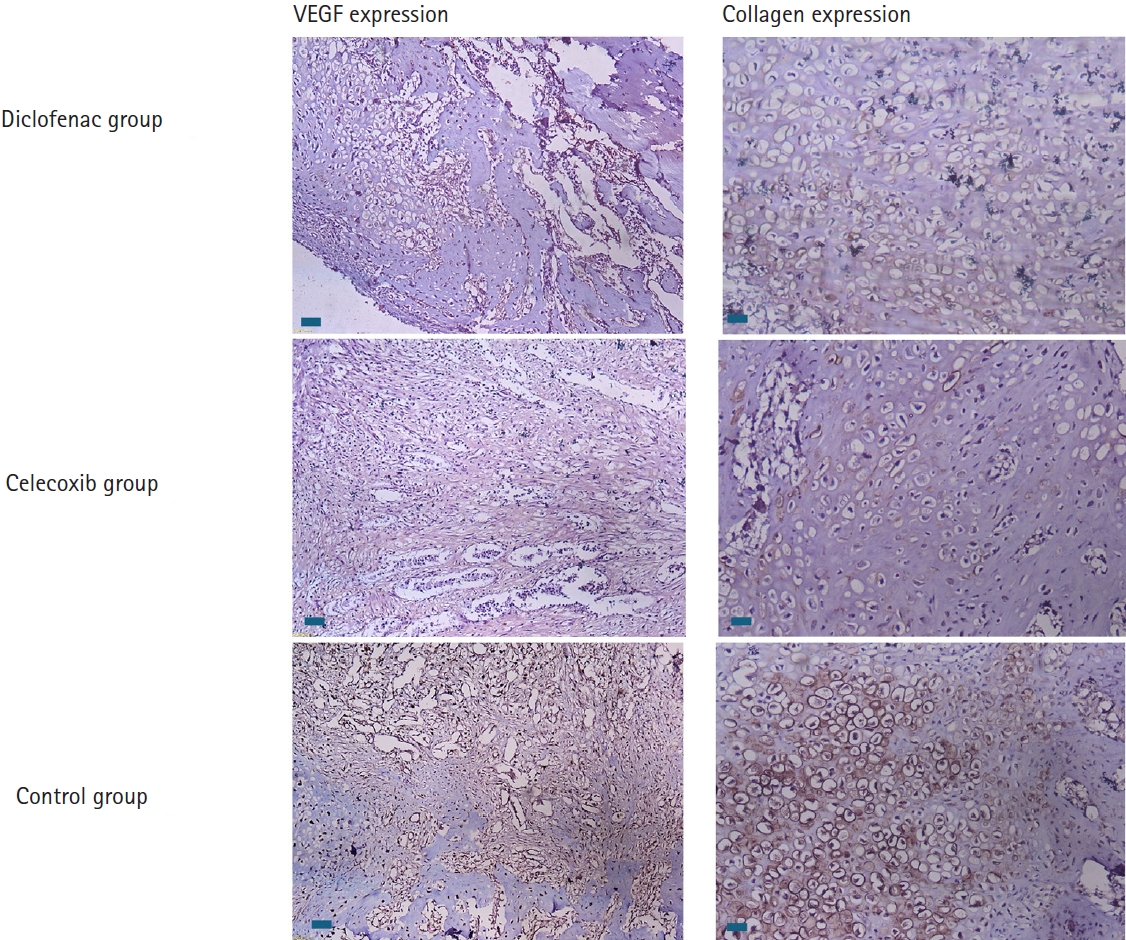
The expression of COLX was less in percentage (P=0.01) and intensity (P=0.02) in experimental animals as compared to controls making IRS significantly lower in these animals (P=0.01) in 2nd week (
Table 3,
Fig. 5). Pairwise comparisons using Dunn’s post hoc test showed significantly lower percentage, intensity, and IRSs in the diclofenac group compared with the celecoxib group (P=0.05, P=0.04, and P=0.04, respectively) and the control group (P=0.003, P=0.009, and P=0.004, respectively). On the 7th week no significant difference was seen in the expression of COLX among animals.
VEGF expression was significantly reduced in experimental animals compared with controls, both in terms of percentage (P=0.005) and intensity (P=0.006), resulting in a significantly lower IRS (P=0.005) at the 2nd week (
Table 3,
Fig. 5). Pairwise comparisons using Dunn’s post hoc test demonstrated significant differences in percentage, intensity, and IRSs between the diclofenac group and controls (P=0.001, P=0.002, and P=0.001, respectively), as well as between the celecoxib group and controls (P=0.04, P=0.04, and P=0.05, respectively).
At the 7th week, VEGF expression showed lower intensity in controls compared with experimental animals (P=0.03), resulting in a significantly higher total IRS in the experimental groups (P=0.03). Pairwise analysis using Dunn’s post hoc test revealed a significant difference only between the celecoxib group and controls for intensity (P=0.03) and IRS (P=0.01).
Correlation between immunohistochemical expression and healing scores
The COLX and VEGF expression was seen to be in significant positive correlation with total healing score (r=0.76, P=0.002; r=0.66, P=0.01 respectively). While in the 7th week they did not show any significant correlation.
Correlation among immunohistochemical expression
Both VEGF and COLX were in significant positive correlation with each other in the 2nd week (r=0.7, P=0.006) while in the 7th week no correlation was seen.
Discussion
Key results
The current study revealed that by the second week, the experimental group receiving diclofenac sodium exhibited a significantly higher fibrosis at the fracture site and delayed bone healing. Furthermore, the expression of COLX and VEGF was notably lower in both experimental groups compared to the control group. By the seventh week, new bone formation was significantly reduced, with a large remaining bone defect between the fractured ends and a significant delay in bone healing in both experimental groups, particularly in the diclofenac sodium-treated animals. Additionally, VEGF expression was decreased in the control group at 7th week likely reflecting advanced healing with reduced angiogenic demand (
Fig. 6). Pairwise comparisons indicate that the most consistent and significant differences are observed between the diclofenac and control groups, whereas celecoxib does not demonstrate uniformly significant differences compared to control across all histological endpoints.
Radiographic examination of the fractured bones at the 7th week showed fracture lines specifically in the experimental animals treated with NSAIDs, which aligns with previous studies [
19,
20]. These findings were further supported by our histological analysis, which revealed larger bone defects and reduced new bone formation at both the 2nd and 7th weeks postoperation, particularly in animals treated with diclofenac sodium. Similar results have been observed in other animal studies, where NSAIDs, especially diclofenac, were found to delay healing by affecting callus remodeling, reducing bone production, and decreasing lamellar bone content in the callus tissue. This occurs due to the inhibition of prostaglandins during the early stages of healing. Prostaglandins, along with various other inflammatory mediators, are known to play a crucial role in osteoblast differentiation [
21-
23].
Despite strong evidence indicating that NSAIDs impair bone healing, a few studies suggest that NSAIDs have little or no effect on fracture healing outcomes [
24,
25]. However, the role of NSAIDs in reducing prostaglandin production, which is essential for regulating bone repair, particularly during the inflammatory phase [
26], may provide the most plausible explanation for the findings in the current study. The inhibition of prostaglandins affects multiple signaling pathways and can delay bone healing by reducing osteoblast activity, increasing osteoclast activity, and impairing angiogenesis.
This explanation accounts for the decreased expression and intensity of COLX and VEGF staining in NSAID-treated animals compared to the control group, particularly at the 2nd week. Several previous studies have reported on VEGF expression in relation to NSAID treatment. For instance, Rofecoxib, a COX-2 inhibitor, has been shown to reduce blood flow and VEGF expression across the fracture gap at 4, 16, and 24 days postinjury in a mouse model [
20]. Additionally, VEGF immunostaining within granulation tissues significantly decreased in animals treated with ketoprofen, a nonselective COX inhibitor, during a four-week study [
27]. NSAIDs are known to impair cell proliferation by inhibiting angiogenesis, as observed in cancer studies, and it is likely that NSAID-induced nonunions occur through a similar mechanism [
20]. However, a study on tibial fractures in mice found that NSAIDs had positive effects on vascular density in the fracture gap during early healing, as compared to the control group [
28]. Nevertheless, this study was unable to clarify the interaction between inflammation and angiogenesis.
Regarding COLX expression, there is a significant lack of data, as no study to date has specifically investigated its expression in relation to NSAIDs. However, it is well established that COLX plays a crucial role in endochondral ossification during fracture healing [
29], as most fractures heal through this process, often involving some degree of cartilage formation. The relationship between COX inhibition and decreased COLX expression has been identified in a few in vitro studies, which suggest that COX inhibition may have negative effects on this process [
30,
31]. COLX is thought to facilitate bone formation during fracture healing by promoting matrix mineralization and angiogenesis. This also explains the linear correlation between the expressions of COLX and VEGF in the current study. Hypertrophic chondrocytes also express VEGF, which promotes vascularization. This process, in turn, accelerates cartilage matrix degradation through the invasion of vessels [
32].
In the current study, at the 7th week, all three groups exhibited similar levels of COLX expression. This aligns with reports indicating that COLX expression peaks around the 14th day, coinciding with chondrocyte hypertrophy. A sharp decline in COLX levels is observed between days 14 and 42, as hypertrophic chondrocytes transition into osteoblasts, and the provisional COLX matrix is replaced by bone [
8]. A similar process likely occurred in this study, which explains the lack of observable differences.
In the 7th week, VEGF expression remained consistent in experimental animals, whereas it decreased in controls. This suggests that bone healing in the fractured femur was not yet complete by the 7th week in experimental animals reflecting the later stages of remodeling, where neovascular support is still required as radiographic and histologic evidence showed the progressive union. As fracture healing transitions from the reparative to the remodeling phase, angiogenic activity does not cease abruptly; instead, it tapers gradually as woven bone is reorganized into lamellar bone. Various processes may influence the interplay between inflammation and angiogenesis. Macrophages, neutrophils, and key bone remodeling cells osteoclasts, osteoblasts, and osteocytes are known to produce angiogenic factors such as VEGF, FGF2, and PDGF, which can regulate directional angiogenesis and maintain the blood supply [
33].
The strong correlation between COLX and VEGF expression and total healing scores in the 2nd week underscores the critical role of both factors in the formation and maintenance of bone tissue during fracture healing. As NSAIDs are known to inhibit the maturation of hypertrophied chondrocytes by blocking COX enzymes, leading to reduced COLX expression and delayed fracture healing [
34], it is evident that COLX expression is essential for enhanced bone formation and improved fracture healing during the early stages. Moreover, reduced VEGF expression can hinder the development of vasculature across the fracture gap, delaying union. Limited VEGF expression disrupts communication between osteoblasts and the vasculature [
35], which likely contributes to delays in the fracture healing process.
This study has certain limitations, the most significant being its nature as an animal-based experimental model; therefore, the findings may not be directly translatable to fracture healing in humans due to interspecies differences in bone biology and healing dynamics. However, it is widely acknowledged that investigating the effects of NSAID administration on bone healing in clinical settings presents substantial methodological challenges, including difficulty in controlling confounding variables and accurately assessing healing outcomes. Therefore, in vivo experimental studies like this one remain a valuable and necessary source of evidence. The prolonged NSAID administration used in this study was intended to assess cumulative effects on fracture healing rather than to replicate routine short-term postoperative analgesic use. Although such exposure may exceed typical perioperative NSAID prescribing, prolonged NSAID use is common in patients with chronic musculoskeletal pain or inflammatory conditions. Nevertheless, extrapolation of dose and duration from animal models to clinical practice should be made with caution.
Implications
In addition, the study focused on histological and molecular endpoints at predefined time points and did not include long-term functional or biomechanical assessments of bone strength. Despite these limitations, the present study enhances our understanding of the biological mechanisms involved in bone repair and offers important insights into the potential influence of NSAID use on the fracture healing process.
Conclusions
Use of nonselective and selective COX-2 NSAIDs delays fracture healing by reducing COLX and VEGF expression in the early stages of fracture healing, which correlates with reduced new bone formation, increased bone defect, and fibrous tissue at the fracture healing site. While our results suggest a potential risk for impaired bone repair, definitive clinical implications cannot be drawn from preclinical data alone. Further studies in humans are needed before firm recommendations can be made.
Article Information
-
Author contributions
Conceptualization: MSI, UZ. Data curation: MZ, RMZ, Formal analysis: UZ. Methodology: MSI, MZ, RMZ. Investigation: MZ, RMZ, ST, MSI. Resources: MZ, RMZ, MSI, AA, UZ. Supervision: UZ. Validation: ST, UZ. Project administration: MSI, AA, UZ. Visualization: MZ, RMZ, ST. Writing-original draft: UZ. Writing-review & editing: MZ, RMZ, ST, MSI, AA, UZ. All authors read and approved the final manuscript.
-
Conflicts of interest
No potential conflict of interest relevant to this article was reported.
-
Funding
None.
-
Data availability
Contact the corresponding author for data availability.
-
Acknowledgments
None.
Supplementary materials
Fig. 1.Lateral radiographs of rat femora at 7 weeks after fracture. (A) The diclofenac group and (B) the celecoxib group show persistent radiolucent fracture lines (white arrows), defined as linear lucencies at the original fracture site indicating incomplete cortical bridging. (C) The control group demonstrates a consolidated bridging callus (red arrow) without residual fracture lucency.
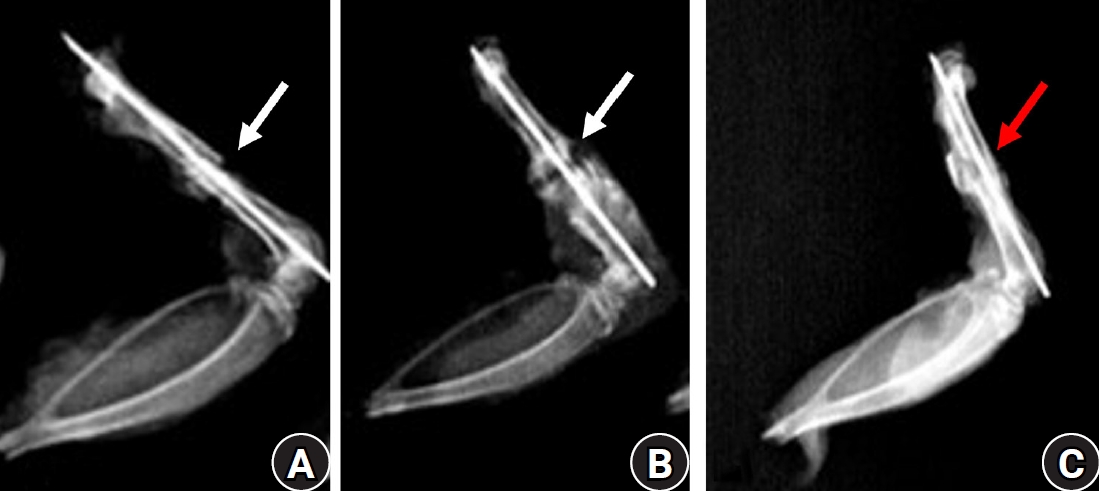
Fig. 2.Photomicrograph of an Alcian blue hematoxylin orange G (ABH/OG)-stained section from the celecoxib group at 2 weeks after fracture, demonstrating cartilage (red arrow), immature woven bone (black arrow), mature lamellar bone (blue arrow), and fibrous connective tissue (yellow arrow) (magnification, ×50).

Fig. 3.Photomicrographs of Alcian blue hematoxylin orange G (ABH/OG)-stained slides illustrating fractured bone edges (*) with callus formation at 2 weeks after fracture in the three animal groups: (A) diclofenac group, (B) celecoxib group, and (C) control group (magnification, ×50). Notably, in the control group, the fracture gap is filled with soft callus tissue (black arrow), unlike in the two experimental groups, where the gap is partially filled with granulation tissue (double-sided and black arrows in A and B, respectively). The bluish staining in C indicates the presence of more cartilaginous tissue.
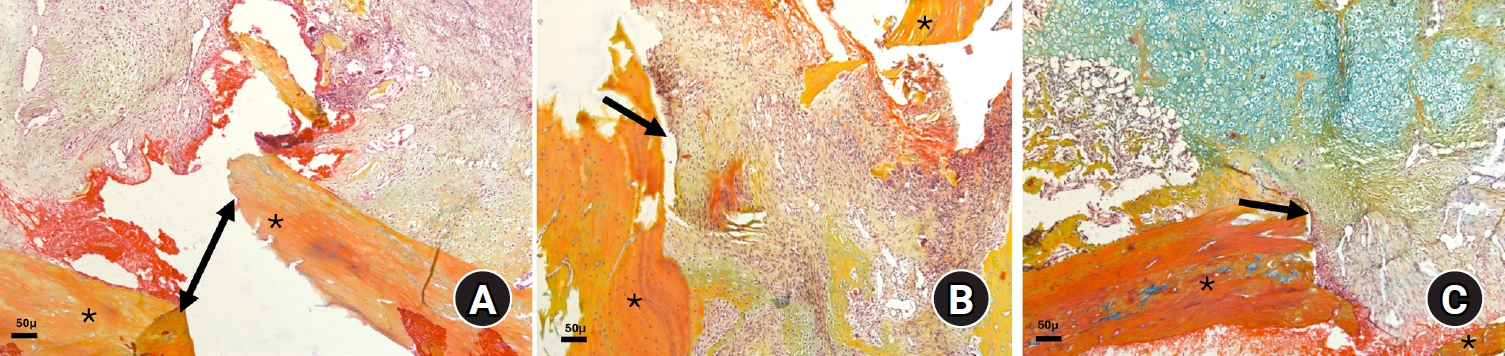
Fig. 4.Photomicrographs of Alcian blue hematoxylin orange G (ABH/OG)-stained slides illustrating fractured bone at 7 weeks after fracture in the three animal groups: (A) diclofenac group, (B) celecoxib group, and (C) control group (magnification, ×50). Notably, in the control group, the bone is almost completely healed (black arrow), whereas granulation tissue is still present in both the diclofenac and celecoxib groups (black arrows). The edges of the healing bone are also visible (*).

Fig. 5.Immunohistochemically stained sections showing the expression and intensity of vascular endothelial growth factor (VEGF) and type X collagen in all three groups at 2 weeks after fracture. Notably, expression is higher in the control group, as shown by the brown staining (scale bars, 50 µm; magnification, ×100).
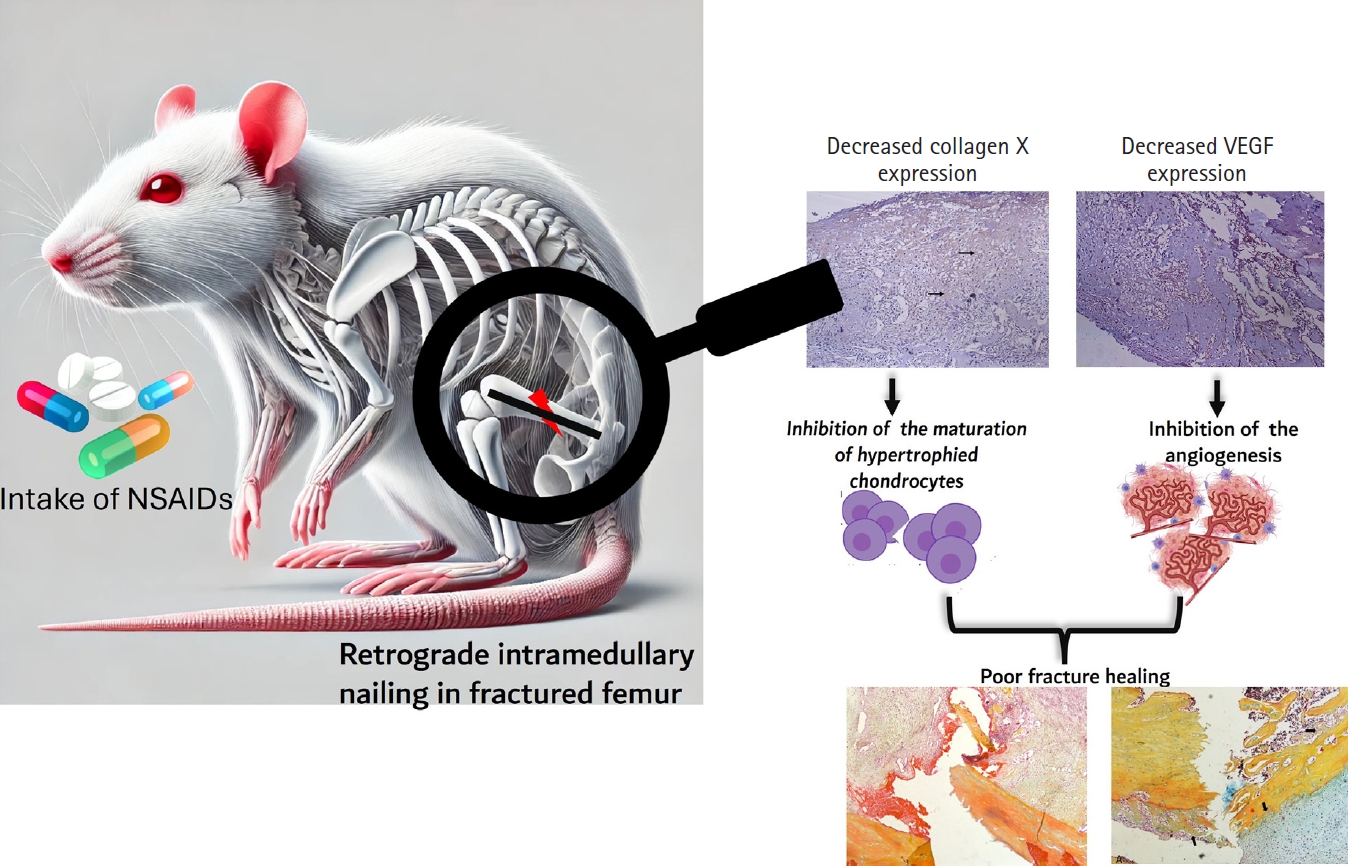
Fig. 6.Intake of NSAIDs reduce the expression of collagen X and vascular endothelial factors (VEGF) in early phase of rat femoral fracture healing. NSAIDs, anti-inflammatory drugs; VEGF, vascular endothelial growth factor.
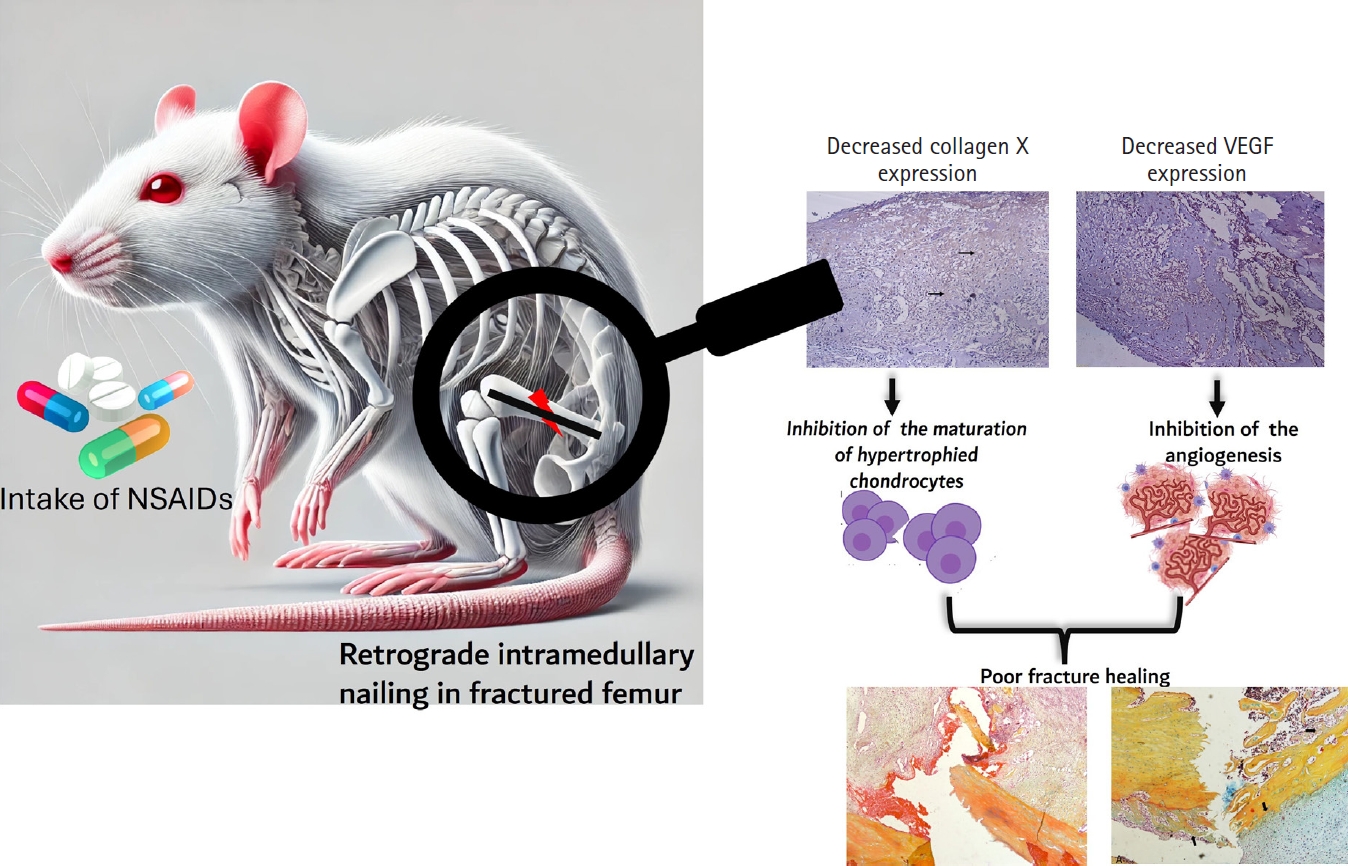
Table 1.Grouping of experimental animals, method of treatment, and drug dosage
|
Group |
Intervention |
Frequency and route of administration |
Duration (wk) |
No. of animals and day of sacrifice |
|
A (n=11) |
Diclofenac sodium 5 mg/kg in 1 mL of distilled water |
Once daily orally from postoperative day 1 until the day before euthanasia |
2 |
5 on PD 15 |
|
7 |
6 on PD 49 |
|
B (n=12) |
Celecoxib 4 mg/kg in 1 mL of distilled water |
Once daily orally from postoperative day 1 until the day before euthanasia |
2 |
6 on PD 15 |
|
7 |
6 on PD 49 |
|
C (n=11) |
1 mL of distilled water |
Once daily orally from postoperative day 1 until the day before euthanasia |
2 |
5 on PD 15 |
|
7 |
6 on PD 49 |
Table 2.Median and IQR of histological scores according to the modified Lane and Sandhu scoring method at weeks 2 and 7 in all three groups
|
Variable |
Group |
Cartilage |
Bone formed |
MHC |
Fibrosis |
Bone defect |
Total |
|
Second week |
Diclofenac |
3 (2–4.5) |
1.5 (1–2) |
5 (4.5–5.5) |
5 (4.5–5.5)* |
8 (3.5–9) |
22 (17–25)* |
|
Celecoxib |
3 (2.5–3) |
2 (1–2.5) |
5 (5–6) |
7 (4.5–7)* |
10 (9–10) |
25 (24.5–27)* |
|
Control |
3.5 (3–5) |
2.5 (1.5–3) |
6 (5.5–6) |
8 (7–8)* |
10 (10–10) |
28.5 (28–30)* |
|
Seventh week |
Diclofenac |
1.5 (1–2.5) |
2 (1.5–2.5)* |
2 (1–5) |
3.5 (1.5–6.5) |
1.5 (0.5–6)* |
13.5 (9–19)* |
|
Celecoxib |
4 (2–5) |
4 (3–6)* |
6 (5–7) |
7 (3–7) |
10 (6–10)* |
31 (21.5–33)* |
|
Control |
1 (1–5) |
6 (5–8.5)* |
6 (1–7.5) |
7 (6–9) |
10 (10–10)* |
31 (29–34)* |
Table 3.Comparison of the median and IQR values for the immunohistochemical expression of VEGF and type X collagen in terms of percentage, intensity, and IRS
|
Variable |
Groups |
Immunomarker expression |
Percentage scores |
Intensity scores |
Total scores |
|
|
Week 2 |
Diclofenac |
VEGF |
1 (0.5–1.5)** |
1 (0.5–1) ** |
1 (0.5–1.5) ** |
|
|
Collagen X |
2 (1.5–2)* |
1 (1–1.5)* |
2 (1.5–3)* |
|
|
Celecoxib |
VEGF |
2 (2–2)** |
2 (1–2)** |
4 (2–4)** |
|
|
Collagen X |
3 (2.5–3)* |
2 (2–3)* |
6 (5–7.5)* |
|
|
Controls |
VEGF |
3 (3–3.5)** |
3 (3–3)** |
9 (9–10.5)** |
|
|
Collagen X |
3.5 (3–4)* |
3 (2.5–3)* |
9 (8.5–10.5)* |
|
|
Week 7 |
Diclofenac |
VEGF |
2 (1.5–2) |
2 (1.5–2)* |
3 (2–4)* |
|
|
Collagen X |
1 (1–1.5) |
1 (1–1.5) |
1.5 (1–2) |
|
|
Celecoxib |
VEGF |
3 (1.5–3) |
2 (1.5–2.5)* |
6 (2.5–7.5)* |
|
|
Collagen X |
2 (1.5–2.5) |
2 (1.5–2.5) |
4 (2.5–6) |
|
|
Controls |
VEGF |
1 (1–1.5) |
1(1-1)* |
1 (1–1.5)* |
|
|
Collagen X |
2 (1–2) |
1.5 (1–2) |
3 (1–4) |
|
References
- 1. GBD 2019 Fracture Collaborators. Global, regional, and national burden of bone fractures in 204 countries and territories, 1990-2019: a systematic analysis from the Global Burden of Disease Study 2019. Lancet Healthy Longev 2021;2:e580-92.ArticlePubMedPMC
- 2. Salari N, Ghasemi H, Mohammadi L, et al. The global prevalence of osteoporosis in the world: a comprehensive systematic review and meta-analysis. J Orthop Surg Res 2021;16:609.ArticlePubMedPMCPDF
- 3. Marsell R, Einhorn TA. The biology of fracture healing. Injury 2011;42:551-5.ArticlePubMedPMC
- 4. Bahney CS, Zondervan RL, Allison P, et al. Cellular biology of fracture healing. J Orthop Res 2019;37:35-50.ArticlePubMedPDF
- 5. Papachristou DJ, Georgopoulos S, Giannoudis PV, Panagiotopoulos E. Insights into the cellular and molecular mechanisms that govern the fracture-healing process: a narrative review. J Clin Med 2021;10:3554.ArticlePubMedPMC
- 6. Sheen JR, Mabrouk A, Garla VV. Fracture healing overview. In: StatPearls. Treasure Island (FL): StatPearls Publishing; 2023.PDF
- 7. Hu K, Olsen BR. The roles of vascular endothelial growth factor in bone repair and regeneration. Bone 2016;91:30-8.ArticlePubMedPMC
- 8. Working ZM, Morris ER, Chang JC, et al. A quantitative serum biomarker of circulating collagen X effectively correlates with endochondral fracture healing. J Orthop Res 2021;39:53-62.ArticlePubMedPDF
- 9. Al Farii H, Farahdel L, Frazer A, Salimi A, Bernstein M. The effect of NSAIDs on postfracture bone healing: a meta-analysis of randomized controlled trials. OTA Int 2021;4:e092.ArticlePubMedPMC
- 10. Ozturan YA, Akin I. Comprehensive review of the impact of NSAIDs on bone healing outcomes in animal models: conflicting evidence and methodological considerations. Discov Med 2024;1:20.ArticlePDF
- 11. Murphy PB, Kasotakis G, Haut ER, et al. Efficacy and safety of non-steroidal anti-inflammatory drugs (NSAIDs) for the treatment of acute pain after orthopedic trauma: a practice management guideline from the Eastern Association for the Surgery of Trauma and the Orthopedic Trauma Association. Trauma Surg Acute Care Open 2023;8:e001056.ArticlePubMedPMC
- 12. Chakkalakal DA, Strates BS, Mashoof AA, et al. Repair of segmental bone defects in the rat: an experimental model of human fracture healing. Bone 1999;25:321-32.ArticlePubMed
- 13. De Giacomo A, Morgan EF, Gerstenfeld LC. Generation of closed transverse fractures in small animals. Methods Mol Biol 2014;1130:35-44.ArticlePubMedPMC
- 14. Udegbunam R, Okereke H, Udegbunam S. Single versus repeated tramadol injection in laparotomized albino rats: comparison of effects on hematology, serum biochemical parameters, and body weight gain. J Adv Vet Anim Res 2015;2:316.Article
- 15. Abbas SS, Schaalan MF, Bahgat AK, El-Denshary ES. Possible potentiation by certain antioxidants of the anti-inflammatory effects of diclofenac in rats. ScientificWorldJournal 2014;2014:731462.ArticlePubMedPMCPDF
- 16. Simon AM, O'Connor JP. Dose and time-dependent effects of cyclooxygenase-2 inhibition on fracture-healing. J Bone Joint Surg Am 2007;89:500-11.ArticlePubMed
- 17. Gultekin A, Unal M, Unlu M, Satoglu S. Effect of human amniotic fluid and membrane on fracture healing on rat fracture model. Med J Suleyman Demirel Univ 2020;27:105-12.Article
- 18. Fedchenko N, Reifenrath J. Different approaches for interpretation and reporting of immunohistochemistry analysis results in the bone tissue: a review. Diagn Pathol 2014;9:221.ArticlePubMedPMCPDF
- 19. Li KH, Cheng L, Zhu Y, Deng GB, Long HT. Effects of a selective cyclooxygenase-2 inhibitor (celecoxib) on fracture healing in rats. Indian J Orthop 2013;47:395-401.ArticlePubMedPMCPDF
- 20. Murnaghan M, Li G, Marsh DR. Nonsteroidal anti-inflammatory drug-induced fracture nonunion: an inhibition of angiogenesis? J Bone Joint Surg Am 2006;88 Suppl 3:140-7.ArticlePubMed
- 21. Beck A, Krischak G, Sorg T, et al. Influence of diclofenac (group of nonsteroidal anti-inflammatory drugs) on fracture healing. Arch Orthop Trauma Surg 2003;123:327-32.ArticlePubMedPDF
- 22. Sandberg O, Aspenberg P. Different effects of indomethacin on healing of shaft and metaphyseal fractures. Acta Orthop 2015;86:243-7.ArticlePubMed
- 23. Menger MM, Stief M, Scheuer C, et al. Diclofenac, a NSAID, delays fracture healing in aged mice. Exp Gerontol 2023;178:112201.ArticlePubMed
- 24. Matsumoto MA, De Oliveira A, Ribeiro Junior PD, Nary Filho H, Ribeiro DA. Short-term administration of non-selective and selective COX-2 NSAIDs do not interfere with bone repair in rats. J Mol Histol 2008;39:381-7.ArticlePubMedPDF
- 25. Inal S, Kabay S, Cayci MK, et al. Comparison of the effects of dexketoprofen trometamol, meloxicam and diclofenac sodium on fibular fracture healing, kidney and liver: an experimental rat model. Injury 2014;45:494-500.ArticlePubMed
- 26. Al-Waeli H, Reboucas AP, Mansour A, Morris M, Tamimi F, Nicolau B. Non-steroidal anti-inflammatory drugs and bone healing in animal models-a systematic review and meta-analysis. Syst Rev 2021;10:201.ArticlePubMedPMCPDF
- 27. Elgendy M, Elsayad G, Seleim M, et al. Flunixin meglumine enhanced bone fracture healing in rabbits associated with activation of early collagen deposition and enhancement of vascular endothelial growth factor expression. Animals (Basel) 2021;11:2834.ArticlePubMedPMC
- 28. Lu C, Xing Z, Wang X, Mao J, Marcucio RS, Miclau T. Anti-inflammatory treatment increases angiogenesis during early fracture healing. Arch Orthop Trauma Surg 2012;132:1205-13.ArticlePDF
- 29. Grant WT, Wang GJ, Balian G. Type X collagen synthesis during endochondral ossification in fracture repair. J Biol Chem 1987;262:9844-9.ArticlePubMed
- 30. Welting TJ, Caron MM, Emans PJ, et al. Inhibition of cyclooxygenase-2 impacts chondrocyte hypertrophic differentiation during endochondral ossification. Eur Cell Mater 2011;22:420-36.ArticlePubMed
- 31. Janssen MP, Caron MM, van Rietbergen B, et al. Impairment of the chondrogenic phase of endochondral ossification in vivo by inhibition of cyclooxygenase-2. Eur Cell Mater 2017;34:202-16.ArticlePubMed
- 32. Kodama J, Wilkinson KJ, Iwamoto M, Otsuru S, Enomoto-Iwamoto M. The role of hypertrophic chondrocytes in regulation of the cartilage-to-bone transition in fracture healing. Bone Rep 2022;17:101616.ArticlePubMedPMC
- 33. Chim SM, Tickner J, Chow ST, et al. Angiogenic factors in bone local environment. Cytokine Growth Factor Rev 2013;24:297-310.ArticlePubMed
- 34. Cottrell JA, O'Connor JP. Pharmacological inhibition of 5-lipoxygenase accelerates and enhances fracture-healing. J Bone Joint Surg Am 2009;91:2653-65.ArticlePubMed
- 35. Hu K, Olsen BR. Osteoblast-derived VEGF regulates osteoblast differentiation and bone formation during bone repair. J Clin Invest 2016;126:509-26.ArticlePubMedPMC










 E-submission
E-submission KOTA
KOTA TOTA
TOTA TOTS
TOTS
 ePub Link
ePub Link Cite
Cite