Articles
- Page Path
- HOME > > Ahead-of print articles > Article
-
Review Article
- Definitive fixation for traumatic pelvic ring injuries: a dynamically informed, posterior-referenced framework
-
Jeong-Hyun Koh
 , Seungyeob Sakong
, Seungyeob Sakong
-
DOI: https://doi.org/10.12671/jmt.2026.00045
Published online: March 24, 2026
Department of Orthopedic Surgery, Ajou University School of Medicine, Suwon, Korea
- Correspondence to: Seungyeob Sakong Department of Orthopedic Surgery, Ajou University School of Medicine, 164 Worldcup-ro, Yeongtong-gu, Suwon 16499, Korea Tel: +82-31-219-5220 Email: sgsy4040@gmail.com
© 2026 The Korean Orthopaedic Trauma Association
This is an Open Access article distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0/) which permits unrestricted noncommercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
- 68 Views
- 2 Download
- Abstract
- Introduction
- Shared language: terms that map to decisions
- Static imaging: necessary but not sufficient
- Deconstructing the “2.5 cm myth”
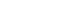
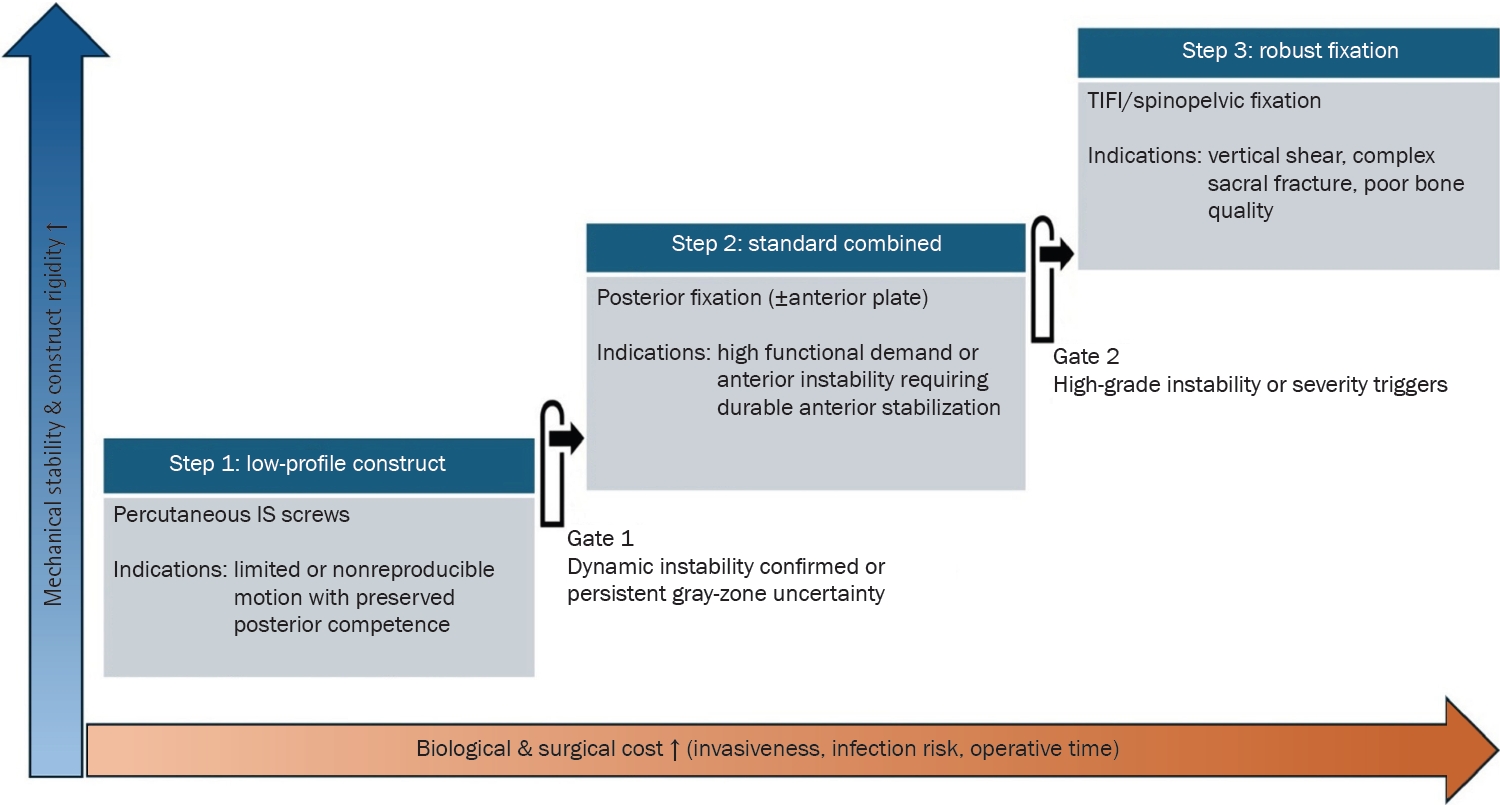
- Dynamic instability assessment: resolving decision-changing uncertainty
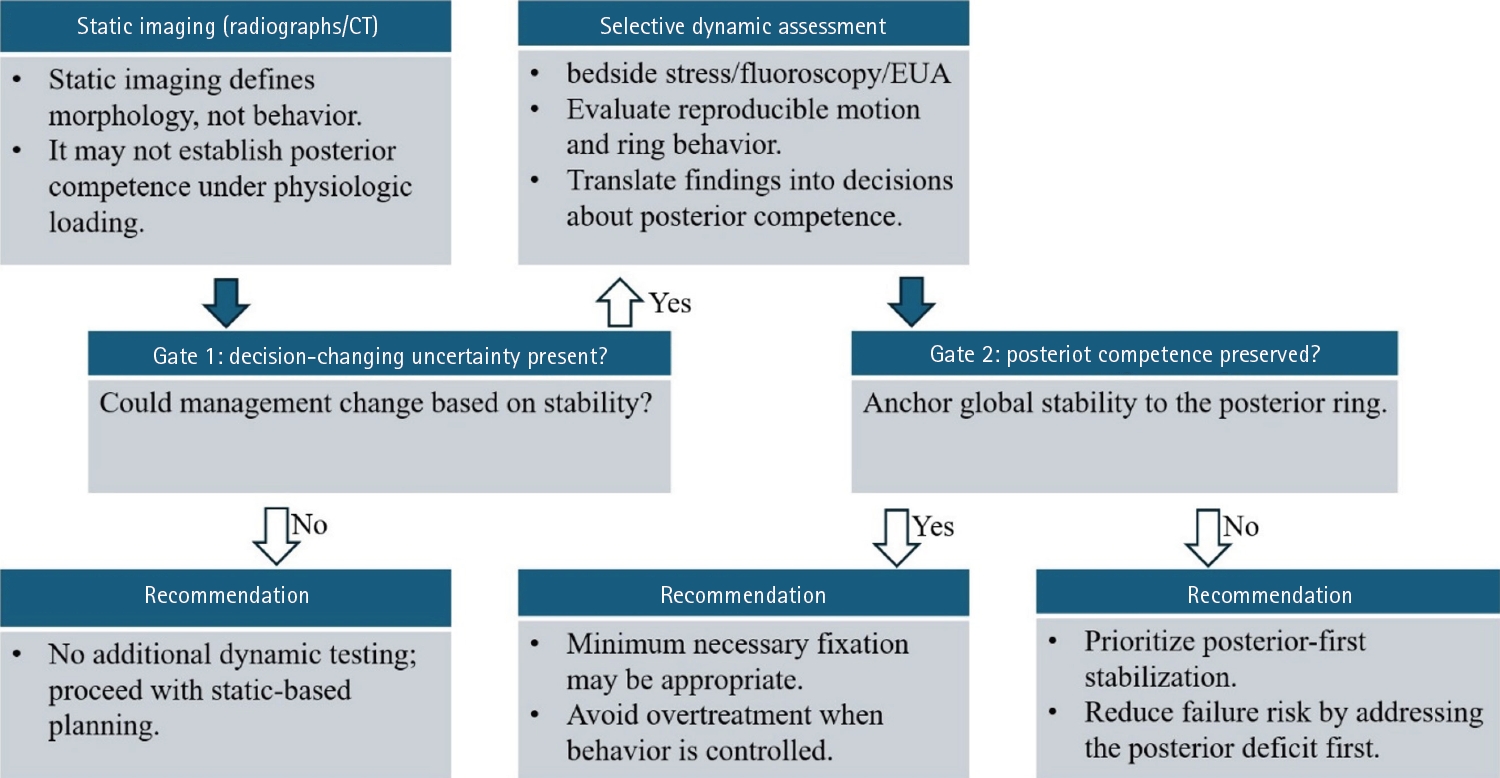
- Translating instability into fixation: escalation along a continuum
- Definitive fixation strategy and sequencing
- Special scenarios and failure prevention
- Conclusions
- Article Information
- References
Abstract
- Optimal definitive fixation for traumatic pelvic ring injuries remains challenging because static radiographs and computed tomography, although essential for defining morphology, do not consistently predict load-dependent behavior during early mobilization. This uncertainty contributes to substantial practice variation and continued reliance on simplified displacement thresholds, such as the 2.5 cm rule. Such rules can misclassify instability by underrepresenting posterior competence and by privileging static measurements over functional behavior. In this narrative review, we propose a dynamically informed, posterior-referenced framework composed of three linked elements: (1) decision-linked terminology that explicitly distinguishes dynamic instability, radiographic change, and clinical failure; (2) selective stress-based assessment when uncertainty is likely to alter management; and (3) escalation along a fixation continuum that weighs incremental stability against operative burden. When static imaging cannot establish posterior competence with confidence, we outline selective stress-based approaches to assess pelvic ring behavior and to translate demonstrated instability into fixation selection along a defined continuum. Across all steps, the framework emphasizes minimum necessary fixation and explicitly incorporates the cost of selection as a primary decision variable. The operative question, therefore, shifts from gap width alone to clinically relevant motion and preservation of posterior competence. In doing so, this approach aims to reduce both undertreatment and overtreatment and to improve the consistency and defensibility of definitive fixation strategies across diverse practice environments.
Introduction
Shared language: terms that map to decisions
Static imaging: necessary but not sufficient
Deconstructing the “2.5 cm myth”
Dynamic instability assessment: resolving decision-changing uncertainty
Translating instability into fixation: escalation along a continuum
Definitive fixation strategy and sequencing
Special scenarios and failure prevention
Conclusions
-
Author contributions
Conceptualization: JHK; SS. Methodology: JHK; SS. Investigation: JHK. Resources: JHK. Data curation: JHK. Supervision: SS. Project administration: SS. Visualization: JHK. Writing–original draft: JHK. Writing–review & editing: JHK, SS. All authors read and approved the final manuscript.
-
Conflicts of interest
No potential conflict of interest relevant to this article was reported.
-
Funding
None.
-
Data availability
Not applicable.
-
Acknowledgments
None.
-
Supplementary materials
None.
Article Information
| Term | Working definition (current review) | Clinical/surgical implications | Key references |
|---|---|---|---|
| Dynamic instability | Clinically meaningful, demonstrable motion/displacement under stress or early physiologic loading, regardless of initial static alignment. | Shifts decision-making from static diastasis to load-dependent behavior in borderline patterns. | [1,2] |
| Radiographic change | Interval widening or minor alignment change after treatment without parallel symptom or functional deterioration. | Discourages reactive escalation driven by imaging alone, particularly for anterior constructs. | [6,7] |
| Clinical failure | Patient-centered deterioration with progressive malalignment that compromises the posterior ring reference frame, persistent disabling symptoms, and/or unplanned revision. | Defines reoperation thresholds by mechanical/functional deterioration rather than radiographic change alone. | [6-8] |
| Gray-zone injury | Presentations in which static imaging is insufficient to classify stability with confidence and management could reasonably differ. | Identifies cases where selective dynamic assessment or structured surveillance is most defensible. | [2,9] |
| Posterior competence | Functional integrity of the posterior ring (posterior sacroiliac complex/ligamentous integrity) as the primary determinant of global load transfer. | Supports posterior stabilization when posterior competence is compromised or cannot be established confidently. | [1,2] |
| Posterior ring reference frame | Interpreting anterior findings and postoperative change through the context of posterior alignment and stability. | Avoids overbuilding the anterior ring to compensate for posterior deficits and reduces radiograph-driven overreaction. | [1,2] |
| Cost of selection | Incremental burden of escalation (operative time, invasiveness, soft-tissue risk, complications, morbidity). | Requires explicit mechanical justification for construct expansion and reinforces “minimum necessary fixation.” | [3,10,11] |
| Minimum necessary fixation | The least extensive construct that reliably controls demonstrated instability and supports safe mobilization. | Anchors escalation: stabilize what is unstable; escalate only when standard constructs are unlikely to suffice. | [1,3] |
Terms are defined to link language to management decisions. Dynamic instability and postoperative anterior findings are interpreted within the posterior ring reference frame, with radiographic change distinguished from clinical failure to avoid imaging-driven escalation. Posterior competence denotes functional integrity of the posterior sacroiliac complex; cost of selection supports the minimum necessary fixation.
CT, computed tomography.
| Fixation level (continuum) | Typical construct examples | Decision triggers (indications) | Primary “cost of selection” | Key references |
|---|---|---|---|---|
| Structured surveillance | Standardized serial radiographs (AP/inlet/outlet) during early mobilization. | When gray-zone presentations show a stable early course, no evidence of clinically relevant motion, and no progressive malalignment. | Risk of delayed displacement; requires disciplined follow-up and a predefined conversion threshold. | [2,9] |
| Isolated anterior fixation | Symphyseal plating; anterior ramus screw fixation. | When posterior competence is confidently preserved and the anterior injury is the primary driver of symptoms or mobilization limits. | Risk of undertreating occult loss of posterior competence; anterior radiographic change may be overinterpreted as clinical failure. | [1,6,7] |
| Standard posterior fixation | 1–2 Iliosacral screws. | When loss of posterior competence is demonstrated or strongly suspected and is expected to compromise reduction maintenance or safe mobilization. | Technical demand and neurovascular risk; loosening risk in poor bone quality or suboptimal reduction. | [15,16,18] |
| Augmented posterior fixation | Additional SI screws and/or transiliac–transsacral screw options. | When posterior control is borderline and added stability is required to prevent recurrent motion and anterior construct overload. | Increased radiation and operative time; diminishing mechanical returns in high-grade vertical patterns. | [12,15,17] |
| Robust posterior fixation | Transiliac internal fixation. | When there is marked vertical tendency, substantial sacral comminution, compromised corridors, or inability to maintain alignment with screws alone. | Greater soft-tissue burden and implant prominence–related symptoms; higher likelihood of implant-related discomfort. | [10,12] |
| Highest-stability constructs | Lumbopelvic/spinopelvic stabilization. | When spinopelvic dissociation or high-grade vertical shear is present and length and shear control are paramount. | Highest morbidity profile; significant wound complication/infection risk; requires explicit mechanical justification. | [10,11] |
| Anterior adjunct (cross-cutting add-on) | Symphyseal plating; ramus screws (added after posterior stabilization). | When a posterior ring reference frame is secured, add selectively if residual anterior instability limits ring closure or mobilization. | Overreaction to expected anterior radiographic change can drive unnecessary revision; interpret anterior changes within the posterior ring reference frame and reserve revision for clinical failure. | [6-8] |
Indications are expressed as decision triggers rather than static cutoffs and should be interpreted within the posterior ring reference frame. “Isolated anterior fixation” presumes posterior competence; “anterior adjunct” is added selectively after posterior stabilization when residual anterior instability is clinically relevant.
AP, anteroposterior; SI, sacroiliac.
- 1. de Ridder VA, Whiting PS, Balogh ZJ, Mir HR, Schultz BJ. Pelvic ring injuries: recent advances in diagnosis and treatment. OTA Int 2023;6(3 Suppl):e261.ArticlePubMedPMC
- 2. Elsissy JG, Ruckle DE, LeBrun C, Johnson JP. Pelvic ring injuries: stable or not? J Am Acad Orthop Surg 2024;32:99-107.ArticlePubMed
- 3. Halawi MJ. Pelvic ring injuries: surgical management and long-term outcomes. J Clin Orthop Trauma 2016;7:1-6.ArticlePMC
- 4. Gansslen A, Lindahl J, Krappinger D, Lindtner RA, Staresinic M. The myth of 2.5 cm symphyseal diastasis. Arch Orthop Trauma Surg 2025;145:306.ArticlePubMedPMC
- 5. Sawauchi K, Esposito L, Kalbas Y, et al. Evolution of management strategies for unstable pelvic ring injuries over the past 40 years: a systematic review. Patient Saf Surg 2024;18:38.ArticlePubMedPMCPDF
- 6. Collinge C, Archdeacon MT, Dulaney-Cripe E, Moed BR. Radiographic changes of implant failure after plating for pubic symphysis diastasis: an underappreciated reality? Clin Orthop Relat Res 2012;470:2148-53.ArticlePubMedPMC
- 7. Eastman JG, Krieg JC, Routt ML Jr. Early failure of symphysis pubis plating. Injury 2016;47:1707-12.ArticlePubMed
- 8. Wheatley BM, Schorr R, Fuhrman H, et al. Can preoperative radiographs predict hardware complication or fracture displacement after operative treatment of pelvic ring injuries? Injury 2021;52:1788-92.ArticlePubMed
- 9. Keltz E, Keren Y, Jain A, et al. Surgical stabilisation in equivocal pelvic ring injuries: into the grey zone. Injury 2023;54:110887.ArticlePubMed
- 10. Patel S, Ghosh A, Jindal K, Kumar V, Aggarwal S, Kumar P. Spinopelvic fixation for vertically unstable AO type C pelvic fractures and sacral fractures with spinopelvic dissociation: a systematic review and pooled analysis involving 479 patients. J Orthop 2022;29:75-85.ArticlePubMedPMC
- 11. Godolias P, Plumer J, Cibura C, Dudda M, Schildhauer TA, Chapman JR. Posterior pelvic ring injuries, lumbosacral junction instabilities and stabilization techniques for spinopelvic dissociation: a narrative review. Arch Orthop Trauma Surg 2024;144:1627-35.ArticlePubMedPDF
- 12. Verbeek DO, Routt ML Jr. High-energy pelvic ring disruptions with complete posterior instability: contemporary reduction and fixation strategies. J Bone Joint Surg Am 2018;100:1704-12.ArticlePubMed
- 13. Wright RD Jr. Indications for open reduction internal fixation of anterior pelvic ring disruptions. J Orthop Trauma 2018;32 Suppl 6:S18-23.ArticlePubMed
- 14. Avilucea FR, Archdeacon MT, Collinge CA, Sciadini M, Sagi HC, Mir HR. Fixation strategy using sequential intraoperative examination under anesthesia for unstable lateral compression pelvic ring injuries reliably predicts union with minimal displacement. J Bone Joint Surg Am 2018;100:1503-8.ArticlePubMed
- 15. Hofmann A, Wagner D, Rommens PM. Iliosacral screw osteosynthesis: state of the art. Arch Orthop Trauma Surg 2025;145:122.ArticlePubMedPDF
- 16. Zhou W, Chen J, Pei X, et al. Incidence of and risk factors for screw loosening after iliosacral screw fixation for posterior pelvic ring injury. Orthop Surg 2023;15:1814-22.ArticlePubMedPMC
- 17. Muller F, Fuchtmeier B. A systematic review of the transiliac internal fixator (TIFI) for posterior pelvic injuries. SICOT J 2021;7:40.ArticlePubMedPMC
- 18. Pishnamaz M, Dienstknecht T, Hoppe B, et al. Assessment of pelvic injuries treated with ilio-sacral screws: injury severity and accuracy of screw positioning. Int Orthop 2016;40:1495-501.ArticlePubMedPDF
- 19. Avilucea FR, Whiting PS, Mir H. Posterior fixation of APC-2 pelvic ring injuries decreases rates of anterior plate failure and malunion. J Bone Joint Surg Am 2016;98:944-51.ArticlePubMed
- 20. Alzobi OZ, Alborno Y, Toubasi A, et al. Complications of conventional percutaneous sacroiliac screw fixation of traumatic pelvic ring injuries: a systematic review and meta-analysis. Eur J Orthop Surg Traumatol 2023;33:3107-17.ArticlePubMedPDF
- 21. Grewal IS, Starr AJ. What's new in percutaneous pelvis fracture surgery. Orthop Clin North Am 2020;51:317-24.ArticlePubMed
- 22. Shen L, Xue X, Ping Y, et al. Evolution of the reduction technique for unstable pelvic ring fractures: a narrative review. Eur J Med Res 2025;30:335.ArticlePubMedPMCPDF
- 23. Jordan MC, Fuchs KF, Herath SC, Windolf J, Meffert RH, Neubert A. Do we need another screw? Sacroiliac screw fixation in open-book pelvic ring injuries (APC type II). EFORT Open Rev 2024;9:827-36.ArticlePubMedPMC
References
Figure & Data
REFERENCES
Citations

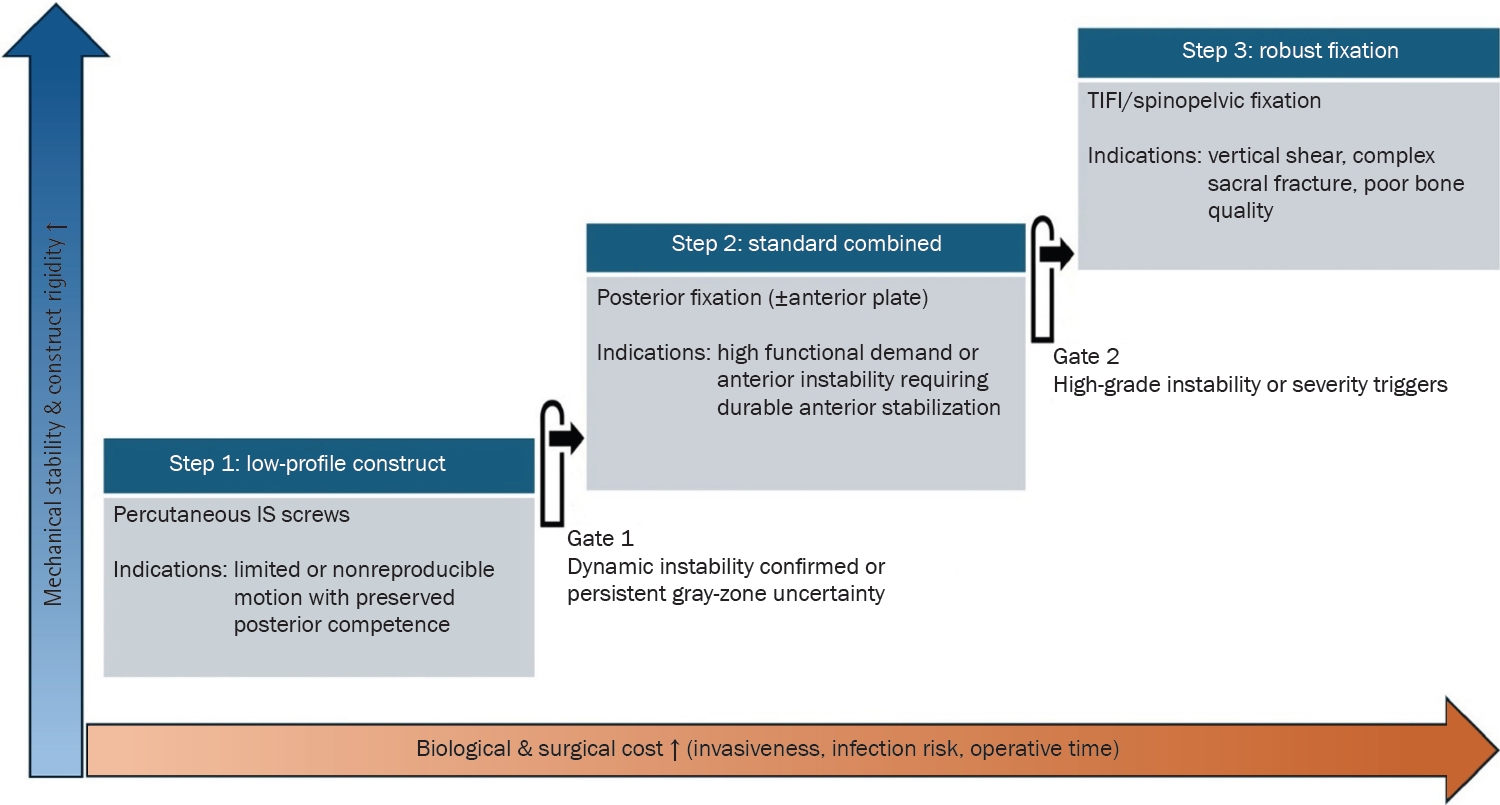
Fig. 1.
Fig. 2.
| Term | Working definition (current review) | Clinical/surgical implications | Key references |
|---|---|---|---|
| Dynamic instability | Clinically meaningful, demonstrable motion/displacement under stress or early physiologic loading, regardless of initial static alignment. | Shifts decision-making from static diastasis to load-dependent behavior in borderline patterns. | [1,2] |
| Radiographic change | Interval widening or minor alignment change after treatment without parallel symptom or functional deterioration. | Discourages reactive escalation driven by imaging alone, particularly for anterior constructs. | [6,7] |
| Clinical failure | Patient-centered deterioration with progressive malalignment that compromises the posterior ring reference frame, persistent disabling symptoms, and/or unplanned revision. | Defines reoperation thresholds by mechanical/functional deterioration rather than radiographic change alone. | [6-8] |
| Gray-zone injury | Presentations in which static imaging is insufficient to classify stability with confidence and management could reasonably differ. | Identifies cases where selective dynamic assessment or structured surveillance is most defensible. | [2,9] |
| Posterior competence | Functional integrity of the posterior ring (posterior sacroiliac complex/ligamentous integrity) as the primary determinant of global load transfer. | Supports posterior stabilization when posterior competence is compromised or cannot be established confidently. | [1,2] |
| Posterior ring reference frame | Interpreting anterior findings and postoperative change through the context of posterior alignment and stability. | Avoids overbuilding the anterior ring to compensate for posterior deficits and reduces radiograph-driven overreaction. | [1,2] |
| Cost of selection | Incremental burden of escalation (operative time, invasiveness, soft-tissue risk, complications, morbidity). | Requires explicit mechanical justification for construct expansion and reinforces “minimum necessary fixation.” | [3,10,11] |
| Minimum necessary fixation | The least extensive construct that reliably controls demonstrated instability and supports safe mobilization. | Anchors escalation: stabilize what is unstable; escalate only when standard constructs are unlikely to suffice. | [1,3] |
| Fixation level (continuum) | Typical construct examples | Decision triggers (indications) | Primary “cost of selection” | Key references |
|---|---|---|---|---|
| Structured surveillance | Standardized serial radiographs (AP/inlet/outlet) during early mobilization. | When gray-zone presentations show a stable early course, no evidence of clinically relevant motion, and no progressive malalignment. | Risk of delayed displacement; requires disciplined follow-up and a predefined conversion threshold. | [2,9] |
| Isolated anterior fixation | Symphyseal plating; anterior ramus screw fixation. | When posterior competence is confidently preserved and the anterior injury is the primary driver of symptoms or mobilization limits. | Risk of undertreating occult loss of posterior competence; anterior radiographic change may be overinterpreted as clinical failure. | [1,6,7] |
| Standard posterior fixation | 1–2 Iliosacral screws. | When loss of posterior competence is demonstrated or strongly suspected and is expected to compromise reduction maintenance or safe mobilization. | Technical demand and neurovascular risk; loosening risk in poor bone quality or suboptimal reduction. | [15,16,18] |
| Augmented posterior fixation | Additional SI screws and/or transiliac–transsacral screw options. | When posterior control is borderline and added stability is required to prevent recurrent motion and anterior construct overload. | Increased radiation and operative time; diminishing mechanical returns in high-grade vertical patterns. | [12,15,17] |
| Robust posterior fixation | Transiliac internal fixation. | When there is marked vertical tendency, substantial sacral comminution, compromised corridors, or inability to maintain alignment with screws alone. | Greater soft-tissue burden and implant prominence–related symptoms; higher likelihood of implant-related discomfort. | [10,12] |
| Highest-stability constructs | Lumbopelvic/spinopelvic stabilization. | When spinopelvic dissociation or high-grade vertical shear is present and length and shear control are paramount. | Highest morbidity profile; significant wound complication/infection risk; requires explicit mechanical justification. | [10,11] |
| Anterior adjunct (cross-cutting add-on) | Symphyseal plating; ramus screws (added after posterior stabilization). | When a posterior ring reference frame is secured, add selectively if residual anterior instability limits ring closure or mobilization. | Overreaction to expected anterior radiographic change can drive unnecessary revision; interpret anterior changes within the posterior ring reference frame and reserve revision for clinical failure. | [6-8] |
Terms are defined to link language to management decisions. Dynamic instability and postoperative anterior findings are interpreted within the posterior ring reference frame, with radiographic change distinguished from clinical failure to avoid imaging-driven escalation. Posterior competence denotes functional integrity of the posterior sacroiliac complex; cost of selection supports the minimum necessary fixation. CT, computed tomography.
Indications are expressed as decision triggers rather than static cutoffs and should be interpreted within the posterior ring reference frame. “Isolated anterior fixation” presumes posterior competence; “anterior adjunct” is added selectively after posterior stabilization when residual anterior instability is clinically relevant. AP, anteroposterior; SI, sacroiliac.

 E-submission
E-submission KOTA
KOTA TOTA
TOTA TOTS
TOTS
 ePub Link
ePub Link Cite
Cite